ISSUE TOPIC: STANDARDISATION AND QUALITY CONTROL OF BIOLOGICALS
SCIENTIFIC RELEVANCE. The use of appropriately certified reference standards (RSs) is a necessary element of analytical procedure standardisation that ensures comparability of results across different laboratories assessing the quality of medicines, which is important for the quality control of biologicals. Marketing authorisation involves providing information on RSs, but no specific requirements are given for the relevant section of the dossier. Therefore, it is necessary to develop the requirements for the appropriate guidelines.
AIM. This study aimed to develop specific requirements for the materials that should be included in the Reference Standards section of the dossier for a biological product.
DISCUSSION. The authors analysed national and international regulatory frameworks governing RSs for biologicals, including documents by the World Health Organisation (WHO), the Eurasian Economic Union, the International Organisation for Standardisation (ISO), and the Russian Federal Agency for Technical Regulation and Metrology. Additionally, the authors analysed the experience of RS certification and use at the Scientific Centre for Expert Evaluation of Medicinal Products. This article covers specific aspects of biological RS certification, considering the differences between measurement techniques and analytical procedures. The authors formulated the key requirements for the documents submitted in the dossier section on RSs for biologicals (biological reference materials). The methods (analytical procedures) used for RS certification should be described in accordance with the requirements for standard operating procedures. Adequate validation studies should establish the necessary validation characteristics of these analytical procedures. Additionally, this article presents the requirements developed to certify primary RSs for structure confirmation of proteins in novel biologicals. The values of certified characteristics and critical quality indicators should be established, and the RS production technology should be described and in compliance with WHO recommendations and/or Good Manufacturing Practice (GMP) standards. The basic certification stages are common to all types of RSs. The RS shelf life should be established in real-time stability studies, whereas accelerated stability testing may be useful for studying degradation mechanisms and the effects of short-term deviations from the specified storage conditions. In accordance with WHO recommendations, the Reference Standards section of the registration dossier should specify changes to the RS that require regulatory approval.
CONCLUSIONS. The authors believe that the requirements for the materials in the Reference Standards section of the dossier for a biological product, as systematised in this study for inclusion in the relevant guidelines, will provide a unified approach to RS development, certification, and documentation for applicants, developers, and competent authority experts and will streamline RS consideration during marketing authorisation.
SCIENTIFIC RELEVANCE. Potency testing of recombinant interferons requires a reference standard. The availability of International Standards (ISs) that are commonly used to assess the quality of recombinant interferons is currently limited. Therefore, the quality of interferons should be assessed using pharmacopoeial reference standards (RSs) certified using ISs (if available).
AIM. This study aimed to certify a pharmacopoeial RS for potency testing of recombinant interferon α-2b.
MATERIALS AND METHODS. The potency determination involved comparing the inhibition of the virus-induced cytopathic effect observed in cell culture with the candidate RS and the WHO International Standard for Human rDNA-derived interferon α-2b. The study used A-549 and MDBK cell lines with encephalomyocarditis (EMC) and vesicular stomatitis (VSV) viruses. The authors followed the requirements of the State Pharmacopoeia of the Russian Federation (Cell-Culture Bioassays for Interferon Products, OFS.1.7.2.0002.15). The results were recorded using instrumental and visual methods. The calculations used mathematical statistics. To factor in the influence of intermediate precision, the authors applied Student’s t-test.
RESULTS. The authors developed a certification programme and procedure and certified the pharmacopoeial RS for recombinant human interferon α-2b (rhIFN α-2b) to identify the corresponding medicinal products (by virus neutralisation) and test their potency (by antiviral activity in cell culture). Upon receipt, the candidate RS was verified for compliance with regulatory specifications. According to the test results, the potency of the candidate pharmacopoeial RS was 4.47×10⁸ IU/mL, and the expanded uncertainty was 8.12×10⁷ IU/mL, with a coverage factor of k=2 and a confidence level of 95%. The pharmacopoeial RS for rhIFN α-2b was considered to have the same shelf-life period as the corresponding medicinal products if stored at a temperature not higher than –40 ºC. When thawed, the pharmacopoeial RS for rhIFN α-2b can be stored at a temperature of 2–8 ºC for up to 1 month.
CONCLUSIONS. Upon certification for potency testing, the pharmacopoeial RS for rhIFN α-2b has been included in the Register of Reference Standards of the Russian Pharmacopoeia as Recombinant Human Interferon α-2b (Potency) FSO.3.2.00455. The introduction of this pharmacopoeial RS will help to conduct the identification and potency testing of all Russian interferon α-2b products at an appropriate scientific and methodological level. As a result, Russian pharmaceutical manufacturers and quality control agencies will no longer depend on imported ISs.
SCIENTIFIC RELEVANCE. An important quality-control issue for therapeutic monoclonal antibodies (mAbs) is the determination of residual protein A leaching from the carrier during the purification of mAbs by affinity chromatography. However, unrelated compounds (immunoglobulins) present in the sample can complicate the immunoenzymatic detection of protein A (matrix effect), potentially leading to false-negative test results. To increase the efficiency of enzyme-linked immunosorbent assay (ELISA), it is necessary to develop a sample preparation step that can irreversibly break the bond in the protein A–mAb complex.
AIM. This study aimed to develop and validate an analytical procedure for the determination of residual protein A in active substances of therapeutic mAbs by ELISA with a test kit comprising in-house reagents.
MATERIALS AND METHODS. Recombinant protein A was used as an antigen. Polyclonal antibodies to protein A were produced by immunising chickens, selecting immunised eggs, and isolating antibodies from these eggs. Protein A-specific antibodies were purified by affinity chromatography. Residual protein A was determined using sandwich ELISA with preliminary sample preparation. The analytical procedure was validated in accordance with the requirements of the State Pharmacopoeia of the Russian Federation (Validation of Analytical Procedures, OFS.1.1.0012).
RESULTS. The authors obtained, isolated, and purified chicken IgY antibodies to recombinant protein A. The authors selected sample preparation conditions for the determination of residual protein A by ELISA and optimum compositions of buffer solutions, including the composition of a denaturing buffer to disrupt the protein A–mAb complex. The developed analytical procedure was validated. According to the measurements of protein A, the accuracy of the analytical procedure ranged within 83–108% of the nominal value, the interlaboratory precision ranged within 96–116%, and the repeatability was up to 13%. The lower limit of quantitation was 10 ng/mL with a minimum required dilution of 1:10. The analytical range extended from 10 to 40 ng/mL. The analytical procedure showed results comparable to those obtained with a similar test kit from an international manufacturer.
CONCLUSIONS. The developed analytical procedure for the determination of residual protein A by ELISA with a test kit comprising in-house reagents can minimise the matrix effect in therapeutic mAbs. This analytical procedure will alleviate import substitution and reduce quality control costs for Russian immunobiologicals.
SCIENTIFIC RELEVANCE. In the quality control of proteolytic enzyme components of medicinal products, the activity of proteases is determined by spectrophotometry, which involves measuring the amidase or esterase activity using a synthetic substrate and the proteolytic activity using the Anson method. These methods require special substrates and have low sensitivity; their specificity may be insufficient, which may lead to serious errors. Quantitative mass spectrometry is an alternative approach to protease activity assays, which involves adding an isotope-labelled peptide to hydrolysates of the test enzyme. This approach allows determining the activity of proteases, notably, by the hydrolysis of specific peptide bonds, while simultaneously confirming the identity and specificity of the test sample. Quantitative mass spectrometry has high sensitivity and does not require special substrates.
AIM. This study aimed to investigate the possibility of enzymatic activity assay and enzyme identification by quantitative mass spectrometry with ¹⁸O labelling through an example of trypsin with casein.
MATERIALS AND METHODS. The study used trypsin, casein, and H₂¹⁸O (Izotop, Russia). Peptide separation was performed using an Agilent 1100 HPLC system; mass spectra were obtained using a Bruker Ultraflex II MALDI-TOF/TOF mass spectrometer. Quantitative mass spectrometry was performed using a standard peptide, which was obtained from casein by tryptic digestion and HPLC purification. For ¹⁸O labelling, the authors dried the peptide and incubated it in H₂¹⁸О water. The quantitative analysis of the product was carried out using MALDI-TOF mass spectrometry. The authors used quantitative mass spectrometry with ¹⁸O labelling to determine enzymatic activity and calculate the Michaelis constant (KM).
RESULTS. Following the tryptic digestion of casein, the authors identified the fragments corresponding to casein chains. The authors produced the isotope-labelled standard peptide and calculated its concentration using mass spectrometry. The authors determined the rate of casein digestion by trypsin and calculated the KM for trypsin, which was 13.65±0.60 μM. The standard deviation for repeated measurements showed that the mass-spectrometric method had a lower error of measurement than the spectrophotometric method. The sensitivity threshold for the mass-spectrometric method was 0.50±0.08 μM.
CONCLUSIONS. The results obtained with trypsin confirm the possibility of enzymatic activity determination by the proposed method of quantitative mass spectrometry with ¹⁸O labelling. According to the sensitivity evaluation results, this method can be used for the simultaneous determination of enzyme activity, identity, and specificity. The proposed mass spectrometry approach is universal, it does not require expensive materials and reagents, and it can be easily adapted to determine the activity of virtually any protease.
SCIENTIFIC RELEVANCE. The sanctions currently imposed on the Russian Federation require
using Russian diagnostic products in vaccine quality assessments, as well as searching for alternative testing methods, in particular, serological ones.
AIM. This study aimed to demonstrate the possibility of using alternative serological methods, including immunoenzymatic, direct agglutination, and indirect haemagglutination methods, for the identification of tularaemia and brucellosis vaccines in studies and quality assessments.
MATERIALS AND METHODS. This study used the established pharmacopoeial reference standard (RS) for the bacterial suspension opacity of 10 international opacity units, the established pharmacopoeial RS for and two commercial batches of the live brucellosis vaccine (Brucella abortus 19 BA), and the established and candidate pharmacopoeial RSs for the live tularaemia vaccine (Francisella tularensis 15 NIIEG). These pharmacopoeial RSs were certified by the Scientific Centre for Expert Evaluation of Medicinal Products. Serological testing used Russian commercial diagnostic products, including dry diagnostic sera (polyvalent brucellosis and tularaemia sera for agglutination tests), a liquid erythrocytic diagnostic preparation of tularaemia immunoglobulin, and enzyme immunoassay (ELISA) diagnostic kits for detecting the causative agents for brucellosis and tularaemia. Statistical analysis involved using Microsoft Excel (P=0.95) for ELISA results and qualitative evaluation for the results of direct agglutination and indirect haemagglutination tests.
RESULTS. All the tested batches demonstrated positive results. Live brucellosis vaccine batches showed positive results in the slide agglutination tests, while live tularaemia vaccine batches yielded positive results in the tube agglutination tests. All indirect haemagglutination tests showed haemagglutination in live tularaemia vaccine samples at the same concentration as positive control samples (6.25×10⁶ cells/mL). ELISA tests showed similar optical density values (D) for the two vaccines and positive control samples. Live tularaemia and brucellosis vaccines (undiluted, 1.0×10⁹ cells/mL) had D=2.133±0.273 and D=0.127±0.013, whereas the corresponding control samples had D=1.942±0.056 and D=0.123±0.007, respectively. The results reflected the presence of brucellosis or tularaemia microbes in the samples, confirming the identity of the vaccines.
CONCLUSIONS. Serological immunoenzymatic, direct agglutination, and indirect haemagglu tination methods with Russian diagnostic products can be used to identify live brucellosis and tularaemia vaccines during quality assessment. The agglutination method with Russian diagnostic sera can be recommended as an alternative quality assessment method for the identification of live brucellosis and tularaemia vaccines, as this method offers time efficiency, simple visual evaluation of results, and low costs and relatively long shelf lives of diagnostic products. However, ELISA and indirect haemagglutination methods cannot be recommended for this purpose because of their labour-intensive and uneconomical nature. The results of this study may support the introduction of the agglutination method in the regulatory documents for live brucellosis and tularaemia vaccines (as an alternative method).
SCIENTIFIC RELEVANCE. The complexity of standardising immunobiologicals, including bacillus Calmette–Guérin (BCG) vaccines, requires improving the approach to statistical analysis of consistency in their quality indicators. Quality analysis tools include Shewhart control charts, which can be used for real-time and retrospective identification and analysis of trends in the processes of interest.
AIM. This study aimed to analyse quality indicators of a BCG vaccine for bladder cancer immunotherapy using Shewhart control charts to monitor consistency in manufacturing processes and quality testing of vaccines.
MATERIALS AND METHODS. The study involved a retrospective analysis of the bacterial concentration, dispersity index, loss-on-drying, and potency data obtained during the quality control of Imuron-vac, a BCG vaccine for bladder cancer immunotherapy, in the past 5 years. As part of the statistical data analysis, the authors calculated the means, investigated the correlation between the test results provided by the manufacturer and the quality control results obtained at the testing centre (TC), and checked the distributions for normality. Individuals and moving range control charts were plotted and analysed for each quality indicator.
RESULTS. The correlation coefficients (r) ranged from 0.34 to 0.70 for the data submitted by the manufacturer and the data obtained at the TC. Pearson’s goodness-of-fit test showed that the distribution of the quality control results obtained at the TC was normal, with χ² = 14.03 for the dispersity index values and χ² = 4.93 for the loss-on-drying measurements. Shewhart control charts for loss-on-drying and potency data suggested that the production process and/or the testing process deviated from the state of statistical control. Consistency monitoring of the bacterial concentration and the dispersity index showed that the state of statistical control was achieved. The authors established control limits for further real-time monitoring of the dispersity index consistency.
CONCLUSIONS. Shewhart control charts are applicable to the quality control of BCG vaccines for bladder cancer immunotherapy at the TC. Continuous monitoring of consistency in quality indicators is needed to improve the standardisation of vaccine production processes and testing methods, as this standardisation will result in high product quality.
RECOMBINANT PROTEINS
SCIENTIFIC RELEVANCE. Pseudomonas aeruginosa is one of the main causative agents of focal and diffuse suppurative inflammations in immunocompromised individuals. The multiple antimicrobial resistance of Р. aeruginosa has created an urgent need to develop effective preventive vaccines against this bacterium and to enhance their protective properties by selecting adjuvants. A promising strategy is to use flagellin, a P. aeruginosa flagellum component inducing the innate immune system through interaction with toll-like receptor 5 (TLR5) and activating the T-cell immune response, as a component or adjuvant in vaccine development.
AIM. This study aimed to produce recombinant flagellin C (FliC) of P. aeruginosa and investigate its immunogenicity, adjuvanticity, and protective properties.
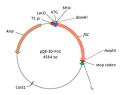
MATERIALS AND METHODS. The fliC nucleotide sequence was obtained by PCR on template DNA of P. aeruginosa PA-103 and was inserted into the pQE-30 plasmid for subsequent expression in Escherichia coli M15. Recombinant FliC purification involved two stages: isolation of inclusion bodies and their dissolution in buffers containing urea and guanidine hydrochloride. Mice were immunised by two intraperitoneal injections with a two-week interval. The immunisation used purified recombinant FliC at a dose of 50 μg per animal and its combination with the Klebsiella pneumoniae surface antigen in a 1:1 ratio. Serum samples from immunised mice were tested for specific antibodies to recombinant FliC by enzyme-linked immunosorbent assay (ELISA). The protective properties of FliC were assessed by intraperitoneal challenge of mice with cultures of P. aeruginosa PA-103 and K. pneumoniae 204.
RESULTS. The authors obtained the producing strain, generated recombinant FliC, and purified the protein to a 97.6% purity. The analysis of serum samples from immunised mice by ELISA and the protection assessment in challenge experiments showed that the purified recombinant FliC protein had immunogenic properties. Furthermore, the experimental challenge of FliC-immunised mice with P. aeruginosa confirmed that recombinant FliC had protective properties, as evidenced by the protection index of 3.0. Recombinant FliC exhibited adjuvant properties, as demonstrated by the effectiveness index of 6.1 observed in the experimental challenge of mice immunised with the combination of recombinant FliC and the surface antigen of K. pneumoniae.
CONCLUSIONS. The purified recombinant FliC protein of P. aeruginosa demonstrated protective activity in mice challenged with P. aeruginosa and adjuvant properties when combined with the K. pneumoniae surface antigen, increasing the immunogenicity of the latter. The use of recombinant FliC holds promise for the creation of a candidate vaccine against P. aeruginosa infection.
BACTERIOPHAGES
SCIENTIFIC RELEVANCE. The incidence of periodontal infections with antibiotic-resistant strains of Actinomyces oris and Aggregatibacter actinomycetemcomitans capable of forming biofilms in the oral cavity is increasing, and the effectiveness of antimicrobials against biofilms is insufficient. Therefore, the isolation of bacteriophages active against A. oris and Ag. actinomycetemcomitans is an urgent task.
AIM. This study aimed to isolate bacteriophages active against A. oris and Ag. actinomycetemcomitans, study their biological properties, and select optimum culture conditions providing pure lines and high titres of bacteriophages.
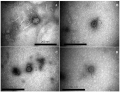
MATERIALS AND METHODS. Bacteriophages were isolated from biosamples of saliva, dental plaque, and periodontal pocket contents. The study determined bacteriophage characteristics, including the optimum multiplicity of infection, phage–cell system cultivation time, resistance to various temperatures and pH, and storage stability. Bacteriophage morphology was studied using transmission electron microscopy. Negative colony morphology, lytic activity, host range, and specificity were assessed using spot testing and the Gratia method. Bacterial strains were obtained from the strain collection of the Micromir research and production centre.
RESULTS. The authors isolated and studied 3 novel bacteriophages active against A. oris (vB_AorP_1/G-12, vB_AorP_2/Ch-28, and vB_AorP_3/Bl-35) and 1 bacteriophage active against Ag. actinomycetemcomitans (vB_AacS_1/Dc-1). The A. oris bacteriophages were classified as podoviruses, and the vB_AacS_1/Dc-1 bacteriophage was classified as a siphovirus. The phages formed completely transparent round plaques without a halo, with a diameter ranging from 0.8±0.1 to 4.0±0.2 mm. The optimum parameters established to obtain pure phages with maximum titres included a multiplicity of infection of 0,1–10 and phage–cell system cultivation time of 8–12 hours. The study demonstrated the ability of A. oris bacteriophages to lyse Actinomyces naeslundii strains. Of the 15 A. oris bacteriophage strains studied, vB_AorP_1/G-12, vB_AorP_2/Ch-28, and vB_AorP_3/Bl-35 lysed 10, 8, and 12 bacterial strains, respectively. The vB_AacS_1/Dc-1 phage isolate exhibited lytic activity against both tested strains of Ag. actinomycetemcomitans. The studied phages demonstrated stability under abiotic stress and long-term storage conditions.
CONCLUSIONS. The authors isolated 3 novel bacteriophages active against A. oris and 1 bacteriophage active against Ag. actinomycetemcomitans and studied their biological properties. The isolated bacteriophages are promising as candidates for further research using clinical strains and whole-genome sequencing.
ISSN 2619-1156 (Online)